Table of Contents
For decades, Africa’s pharmaceutical approvals were slowed by a patchwork of regulatory systems, with each country operating independently. A manufacturer seeking to sell the same product across several countries had to submit separate dossiers, pay multiple fees, respond to repeated scientific queries, and endure approval timelines that could stretch from 12 months in some markets to over 30 in others. Fragmented oversight, uneven technical capacity, and duplicated assessments made multi-country operations costly and uncertain, discouraging large-scale investment and slowing access to medicines for patients across the continent.
The landscape began to change in 2025. The World Health Organization implemented its most significant Prequalification Programme reform in 20 years, streamlining approval timelines and reducing duplication for products eligible for donor-funded procurement.
Simultaneously, the African Medicines Agency moved from treaty ratification to operational status, establishing a single-window pathway that allows manufacturers to submit once for a continental scientific assessment. While national issuance remains with individual states, over 30 countries have now committed to the AMA framework, with lead markets increasingly using regulatory reliance to fast-track local approvals based on the agency’s unified review. Together, these reforms compress time-to-market and reduce administrative burdens that historically slowed revenue realization.
For investors, the consequences are immediate and material. Products that previously generated staggered revenue across multiple years can now enter multiple markets simultaneously, improving cash flow and scaling potential. The speed, predictability, and reduced regulatory friction transform the investment calculus for pan-African pharmaceutical ventures. Companies pursuing pan-African pharmaceutical strategies, such as Uganda’s Dei BioPharma or regional manufacturers like Quality Chemical Industries Limited, illustrate the type of ventures that could benefit from a more unified regulatory pathway as they seek to distribute medicines across multiple African markets. These reforms could make it easier for such projects to scale beyond a single national market, opening opportunities that were previously considered too complex or risky.
How was the African pharmaceutical market before these reforms?
Africa’s pharmaceutical market has long been defined by fragmentation. The push for regional harmonization began in 2009 with the African Medicines Regulatory Harmonization initiative, which aimed to align technical standards across countries. In practice, however, national regulators continued to require separate reviews for each product, forcing manufacturers to submit multiple dossiers, pay multiple fees, and answer the same scientific questions.
By 2020, these duplications translated into significant delays. A full product review could take 240 days in Côte d’Ivoire or 247 days in Nigeria, and timelines in other countries often stretched even longer. Around 90% of national regulatory authorities were considered minimally functional for core regulatory tasks, creating bottlenecks that slowed market entry across the continent. Even products that secured the WHO Prequalification necessary to access donor-funded procurement channels still faced lengthy domestic reviews, further compounding delays.
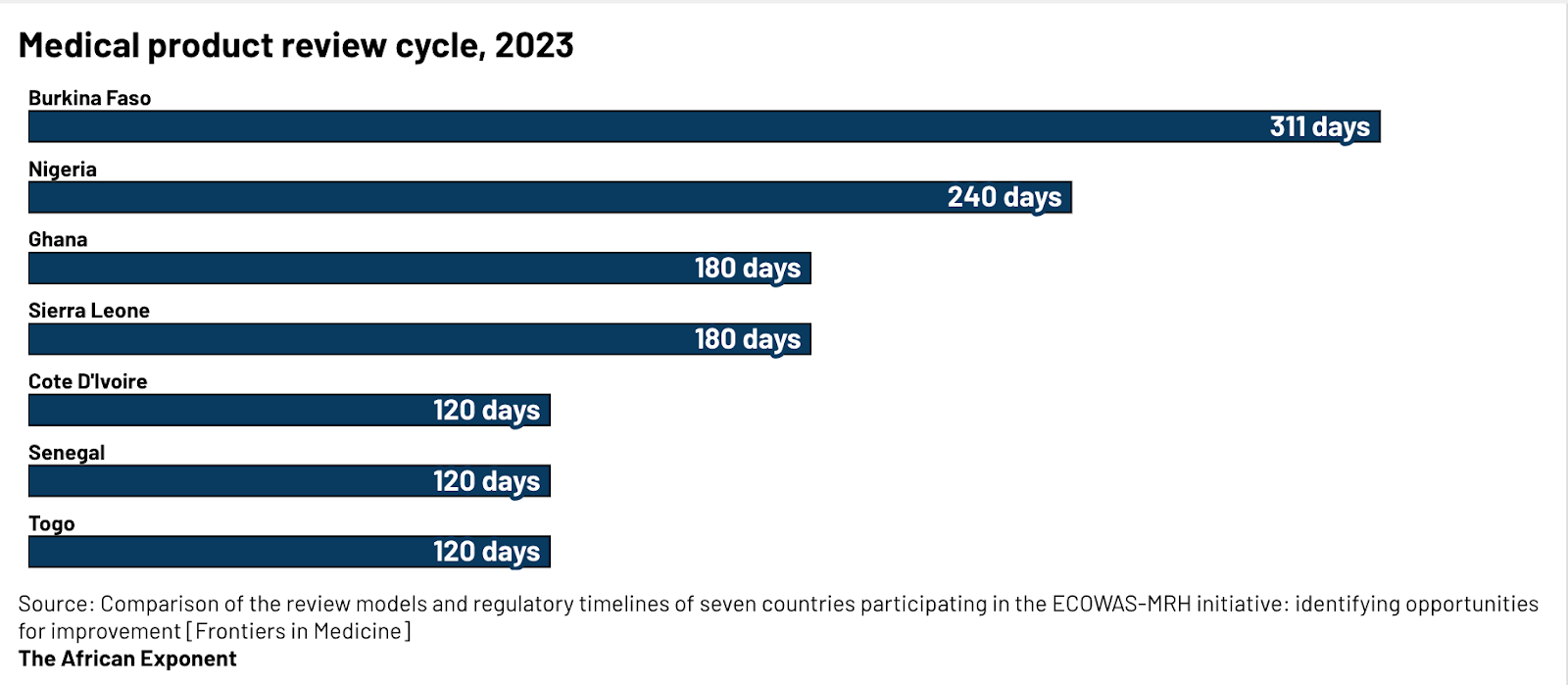
The effects were tangible. Africa’s 375 drug manufacturers produced only 10-30% of the medicines consumed locally, leaving the continent dependent on imports despite rising demand. For investors, this system created what many called a “fragmentation premium,” which includes the cost, delay, and risk built into launching across multiple African markets. Understanding this history is essential to appreciating the significance of the WHO and AMA reforms, which now promise a dramatically faster path to market.
What changed with the WHO Prequalification reform and the AMA single‑window pathway?
Building on the delays and duplication that once defined Africa’s pharmaceutical market, two reforms have sharply altered the landscape. First, the WHO Prequalification Programme underwent its largest overhaul in two decades, streamlining review processes and reducing the steps required for manufacturers to qualify for donor-funded procurement channels. By aligning assessments with regional standards and providing targeted support for African manufacturers, the reform has cut months from the time it takes products to reach markets like PEPFAR, Gavi, and the Global Fund.
Second, the African Medicines Agency (AMA) became fully operational in October 2025, introducing a single-window pathway that allows a product to undergo one continental scientific assessment instead of dozens of separate national reviews. With at least 30 participating member states, AMA now coordinates regulatory approvals, mutual recognition, and inspections across borders, compressing years of fragmented approvals into a single, harmonized process.
The impact is immediate and measurable. Products that previously faced multi-year delays may now benefit from faster and more predictable pathways, depending on national authorities recognizing AMA assessments. Companies like Quality Chemical Industries Limited have historically faced long approvals across multiple markets, while ventures such as Dei BioPharma are actively seeking pan-African distribution. As more countries adopt the AMA framework, these ventures could scale more easily and with less risk.
Early experiences suggest that coordinated reviews could significantly shorten multi‑country timelines in the near future. As Dr Semete‑Makokotlela, Chair of the AMRH Steering Committee, explains, “the procedure… is expected to shorten the registration process from years to mere months or weeks,” highlighting how harmonised assessments can compress approval times across the continent.
Manufacturers can leverage AMA’s framework to align submissions and benefit from coordinated scientific guidance, potentially reducing the need for repeated national reviews. While approvals in each country still depend on national procedures, this harmonized approach is expected to shorten timelines and lower administrative costs compared with the fully fragmented system of the past.
Why does this matter for investment strategy and returns?
For investors, the biggest constraint in African pharmaceutical deals was timing and uncertainty. When approval timelines stretched over years and varied by market, companies could not plan production, sales, or cash flow with confidence. Under the old system, a manufacturer might launch in one country early while waiting on others, meaning revenue did not scale evenly, and working capital stayed tied up in regulatory queues.
While full multi‑country launches under the AMA single‑window system are still emerging, recent WHO Prequalification achievements, such as Swiss Pharma Nigeria Limited’s approval of a zinc sulfate dispersible tablet and industry efforts to pursue harmonised pathways, indicate growing engagement with streamlined regulatory mechanisms. This increases early revenue and reduces the lag between capital deployment and earnings realization. In investment terms, that shift increases net present value (NPV) and improves internal rate of return (IRR) because cash flows occur sooner rather than later.
The change also affects risk assessment. When a company no longer has to budget for repeated submissions and staggered approvals, the cost of getting to market declines, reducing the overall cost of capital. Funds are freed up for production scale‑up and distribution, instead of being locked in compliance cycles. More predictable regulatory timing also allows for clearer financial modeling, which lowers the risk premium investors apply to African pharmaceutical ventures.
In short, these reforms convert a historically slow, unpredictable investment environment into one where returns can be modeled with greater certainty, and capital can be deployed with less risk and greater speed.